The pharmaceutical industry is on the guard of people’s health and well-being. At the same time, it faces numerous complexities, challenges, and risks that can hamper the implementation of the main mission of the domain—making people healthier and saving lives. Read the article to learn the peculiarities of pharmaceutical project management, the key challenges these projects face, and how you can overcome them with a robust project management tool.
Key takeaways:
- Pharmaceutical project management deals with drug discovery, clinical trials, regulatory compliance, drug testing, and manufacturing.
- Pharmaceutical projects are complex, face regulatory compliance difficulties, high risks and uncertainty, long timelines, resource management difficulties, and pressure for fast delivery.
- Detailed project planning, risk and resource management, and cross-departmental collaboration allow pharmaceutical companies to enhance project outcomes and bring drugs to market faster.
- Project management tools like Epicflow help pharmaceutical companies streamline the work on projects, manage resources effectively, address risks, and ensure the success of even the most complex pharmaceutical projects.
Specifics of Pharmaceutical Projects
To be able to manage pharma projects effectively, you should understand their specificity. Pharmaceutical projects are endeavors related to the drug development process, starting from drug discovery to the final approval of a new medication. Let’s take a closer look at the main types of projects the pharmaceutical industry deals with.
Drug discovery and development.
These projects aim to identify candidates for new drugs. They also involve preclinical and clinical trials, testing, and regulatory approvals. Projects of this type involve extensive research and have extended project timelines.
Clinical trials.
This type of pharma projects involves testing newly developed drugs to make sure they are safe, effective, and comply with required regulations. These projects are implemented in phases with increasing complexity and amount of data to analyze.
Regulatory compliance.
The pharmaceutical industry has strict standards and regulations for drugs. So, some pharma projects involve implementing processes and preparing documentation to ensure the newly developed medications comply with all necessary standards.
Drug manufacturing.
These projects deal with the actual production of drugs once they are approved. These projects also involve work coordination at manufacturing facilities, managing material resources and equipment, ensuring quality control, etc.
Read more: Top Trends in the Pharmaceutical Industry [2025]: What to Expect?
Importance of Project Management for Pharmaceutical Industry
Project management is a convenient way to manage various initiatives and endeavors; it helps plan, organize, monitor progress, allocate resources, and manage risks. Let’s review how effective pharma project management optimizes the completion of endeavors in the industry, helps address their complexity and overcome constraints.
Reducing projects’ lead times
Pharma projects are often long-term. Effective project management makes it possible to reduce timelines thanks to increasing resource efficiency as well as eliminating bottlenecks and inefficiencies.
There’s also a unique dimension to lead time improvements in the pharmaceutical industry, saving human life. As Stephen Devaux puts it:
In 2020, the US Federal Emergency Management Agency set the value of a “statistical life” at $7.5 million. The European Union’s member states in 2016 averaged €3.3 million. If a new product will save the life of one European per day, even in 2016 terms that would mean a drag cost of €3.3 million * 7 = €23 million for every week of delay on the critical path activities for such a product.
More effective resource utilization
Intelligent resource management is part and parcel of effective project management. It involves the proper allocation of human and material resources, monitoring and optimization of their utilization. The importance of resource management increases when a company manages multiple concurrent projects or project portfolios.
Read more: Resource Utilization Made Simple: Essence, Importance, and Ways to Optimize It
Effective risk management
Pharmaceutical activities are vulnerable to risks. Risk management is an integral part of the project management process, which helps pharmaceutical companies identify possible threats and opportunities and develop strategies to respond to risk-bearing situations. Proper risk management increases the effectiveness of pharmaceutical activities thanks to eliminating roadblocks and taking advantage of opportunities.
Improved budget management
Drug development and related processes are rather costly. Efficient project management helps pharmaceutical companies monitor and control the budget allocated for these activities, prevent cost overruns by increasing resource efficiency and streamline workflows.
Read more: Optimizing Portfolio Profit Through DIPP-Guided Resource Allocation
Enhanced collaboration
Pharmaceutical activities and processes require collaboration between diverse experts. The project management process contributes to effective communication and collaboration between these specialists and increases the effectiveness of their joint efforts in developing and delivering new drugs.
Despite the numerous benefits of project management for the pharmaceutical industry, it’s sometimes easier said than done. Read the next section to discover the main difficulties of pharmaceutical project management.
Challenges in Managing Pharmaceutical Projects
Understanding the potential challenges of pharmaceutical projects will help you manage them more effectively. These challenges are explained by the highly regulated, time-sensitive, and complex nature of drug development. So, here are the main difficulties you may face in pharma projects.
Project complexity
Pharma projects involve research, regulatory approval, manufacturing process, administering supply chains, etc. These processes can be interdependent and require thorough coordination. In addition, clinical trial projects are characterized by increased complexity, as they require time for patient recruitment, may involve unforeseen pharma industry challenges (e.g., adverse reactions or unforeseen outcomes), have to meet regulatory requirements, are costly, etc.
Compliance with regulations
The need to comply with strict requirements and standards is part and parcel of pharmaceutical projects. This becomes a complexity, as each regulatory body has their own rules for drug development stages, which may result in additional delays and costs. In addition, these requirements are regularly updated, which poses the need for pharmaceutical companies to stay up-to-date with these changing standards.
High risks and uncertainty
Pharmaceutical projects are subjected to various risks: supply chain disruptions, issues during clinical trials, safety concerns, unpredicted effects of new drugs, implementing innovations, etc. These potential risks may lead to significant delays, increased project costs, or even project failure.
Managing distributed project teams
Pharma projects often involve the collaboration of specialists from various fields of knowledge: researchers, regulatory experts, manufacturing professionals, marketers, quality assurance specialists, etc. Project managers should ensure proper communication and collaboration between these experts for the seamless flow of pharmaceutical projects.
Read more: 4 Tips on How to Manage Globally Distributed Teams with Maximum Efficiency
Long duration
Pharma projects, especially when it comes to drug development, can last for years or even decades, which is explained by the need for extensive testing and regulatory approval. This becomes a challenge, as a lot of factors affecting project success can change during this period. At the same time, pharmaceutical companies face severe competition with each other for being first to market. These conflicting factors create a real challenge for pharmaceutical companies and project managers.
The need for faster delivery
Pharmaceutical companies face severe pressure to deliver their projects on time. First, it’s explained by competition with other companies; delayed projects result in significant financial losses and missed opportunities. Second, pharma project delays can lead to patent expiration, which means that a company will no longer have the right to market the drug it has developed. So, despite the complexity and constraints of pharmaceutical projects, managers should make every effort to complete them on time.
Now that the main challenges of pharmaceutical projects are clear, let’s consider how they can be solved with the right software depending on a company’s specific needs. For example, portfolio management tools help pharma companies keep up with drug development complexity and ensure strategic alignment of their projects. PMO software ensures standardization of project management processes across the entire organization. Project resource management software enables staffing projects with required resources and their efficient utilization across projects. Finally, robust capacity planning software helps project leaders forecast possible bottlenecks and plan future projects intelligently.
Project Management in the Pharmaceutical Industry: Case Study
Let’s look at how project management can help a pharmaceutical company improve performance on a concrete example. A large US pharmaceutical company was experiencing the following issues:
- Lack of data on resource capacity.
- Lack of prioritization.
- A large backlog of projects.
- Overloaded resources.
This company turned to Epicflow for help and was able to:
- Import project performance data to Epicflow.
- Visualize future bottlenecks.
- Use intelligent workload management to avoid them.
This allowed the pharmaceutical company to increase output with the same amount of resources and gain a better visibility into its portfolio.
Overcoming Challenges in Pharmaceutical Projects with Project Management Software
Before we consider the opportunities provided by pharmaceutical project management software, we’d like to outline some steps that will help you address the above-mentioned challenges.
- Plan for risks and uncertainties.
- Prevent and eliminate bottlenecks.
- Increase resource efficiency.
- Enhance communication and collaboration.
- Regularly monitor and control project and resource performance.
- Make decisions based on data analysis.
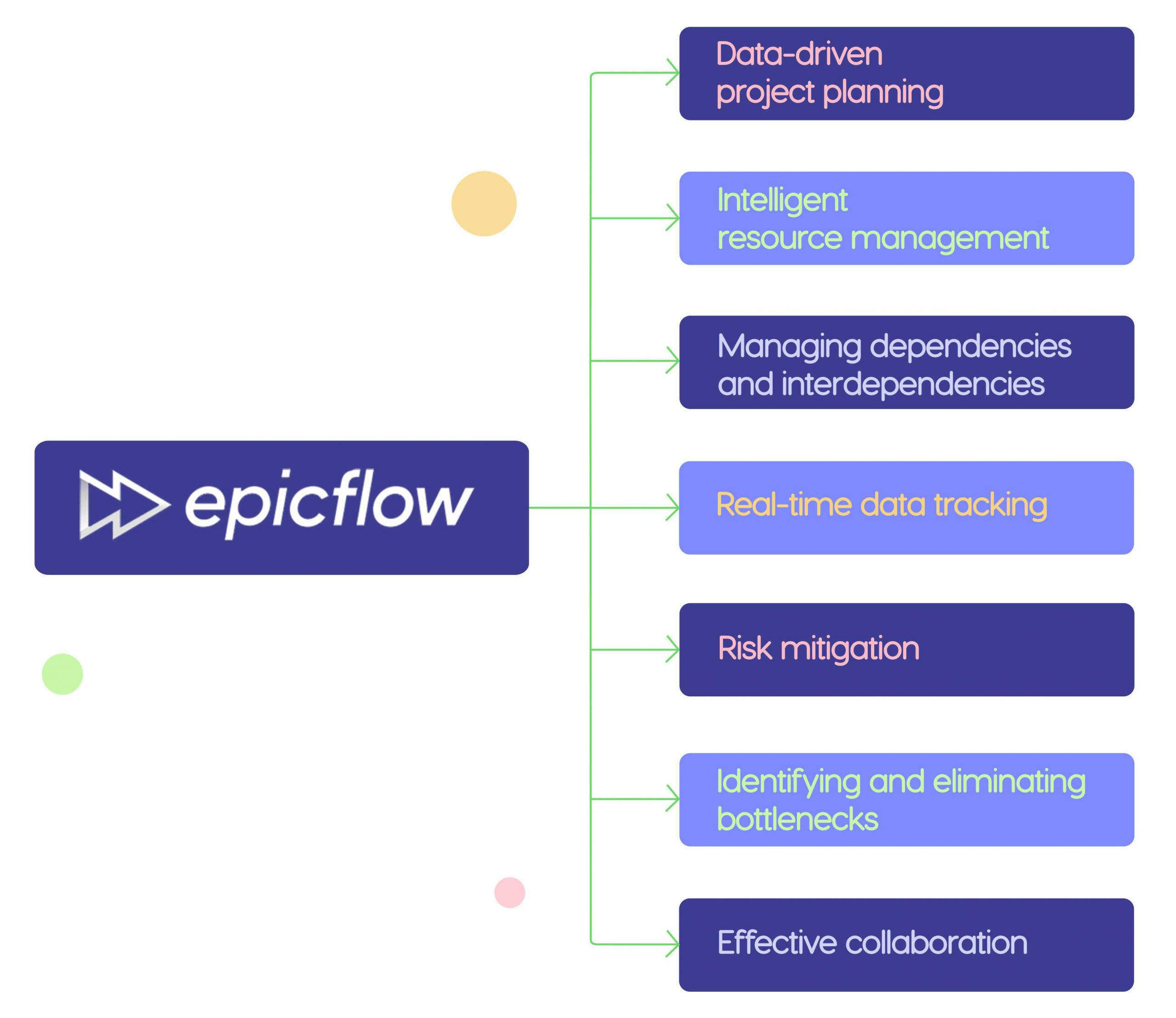
Of course, implementing all these measures will be challenging without the assistance of the right project management tool. So, let’s look at opportunities provided by Epicflow, pharmaceutical project management software designed for managing multiple complex projects with limited resources.
1. Data-driven project planning.
Epicflow makes it possible to create coherent project plans based on forecasts and scenario analysis. As a result, you can develop realistic plans taking into account all existing constraints, which will help you prevent unpleasant surprises in the workflow. For example, before starting a new project, you can assess its feasibility, find out resource capacity, and come up with manageable timelines.
Epicflow’s new feature, EPO, takes this to a new level by introducing AI-powered data-driven project balancing. Given project business value estimations and existing capacity constraints, it will automatically sequence pharmaceutical projects to achieve an optimal throughput.
2. Intelligent resource management.
Epicflow’s AI-powered system optimizes resource allocation: it helps assign resources based on their skills, capacity, and availability, which guarantees that key resources—whether human or material—are available where they are needed most. This helps avoid resource bottlenecks and ensures that all project phases run smoothly.
3. Managing dependencies and interdependencies.
Pharma projects often consist of multiple interrelated phases that may overlap across multiple projects. Epicflow provides a clear overview of all project dependencies and timelines, helping managers monitor the project flow, identify potential bottlenecks, and ensure that critical activities are executed in the correct sequence.
4. Real-time data tracking.
Epicflow’s real-time tracking capabilities provide project managers with immediate insights into project progress, bottlenecks, budget consumption, and resource utilization. This facilitates more effective decision-making and helps managers quickly respond to any issues, e.g., delays in clinical trials, supply chain challenges, etc.
5. Risk mitigation.
Pharmaceutical projects face many risks and uncertainties, from regulatory delays to unexpected side effects in clinical trials. Epicflow’s What-if Analysis feature allows project managers to simulate different scenarios, analyze their consequences, and develop contingency plans, reducing the impact of risk-bearing events.
6. Identifying and eliminating bottlenecks.
Eliminating bottlenecks in the workflow is one of the cornerstones of Epicflow’s approach. Our system has features that can forecast and detect bottlenecks, identify their causes, and offer ways to address them. Moreover, our AI assistant Epica can give early warnings of bottlenecks and suggest measures to eliminate them. With this approach, you can streamline project work, prevent project delays, and significantly increase resource efficiency.
7. Effective collaboration.
Epicflow allows project teams to collaborate effectively. It provides all project participants with a unified platform for tracking tasks, milestones, progress, resource performance, and other project-related information. It also improves resource management in pharmaceutical industry by helping project managers coordinate activities between cross-functional teams and provide every project participant with a single source of truth, which contributes to more effective work on projects.
Therefore, leveraging Epicflow for pharmaceutical projects will help address their complexity, overcome challenges, increase resource efficiency, effectively manage risks, and more. You can book a call with our specialists to learn about more opportunities Epicflow provides to ensure the achievement of project goals by pharmaceutical companies.
Conclusions
The pharmaceutical project management is impacted by the specifics and complexity of the pharmaceutical industry. The vast diversity of projects needed to make a company successful, the different skills needed to execute them, and the constant pressure to push products to market fast make project management in the pharma industry difficult.
Using real-time and historical performance data to inform your strategy and mitigate risks, having clear project priorities, and using PPM software to balance the portfolio can alleviate many of those issues.
Frequently Asked Questions
What is project management in pharmaceutical industry
Project management in the pharmaceutical industry is a complex process that involves project planning, prioritization, and scheduling as well as resource allocation and coordination a to ensure a smooth execution and timely delivery of all projects in a portfolio.
What is the biggest challenge in the pharmaceutical industry?
The biggest challenges in the pharmaceutical industry involve cybersecurity, logistics and supply chain challenges, workforce shortages, and regulatory difficulties.
How to address challenges in pharmaceutical project management?
Leveraging appropriate project management software can help pharmaceutical project and portfolio managers overcome the challenges. For example, a tool like Epicflow can help prioritize projects and tasks, optimize resource utilization, detect and resolve bottlenecks, provide comprehensive control of projects, improve risk management and decision-making, and facilitate effective collaboration.
Why is project portfolio management particularly challenging in the pharmaceutical industry?
Pharmaceutical project and portfolio management is challenging due to the complexity of pharmaceutical projects, high risks and uncertainty, long duration and the need for faster delivery as well as strict regulatory requirements.
What are best practices for integrating clinical trials into pharmaceutical project management?
Clinical trials combine commercial and scientific vision of a pharmaceutical company with regulatory requirements, which makes them uniquely difficult. To integrate them into project management effectively, you need to create contingency plans for regulatory risks and especially for the risk of increased approval timelines.
What project management methodologies are commonly used in the pharmaceutical sector?
The pharmaceutical industry primarily uses project management methodologies like waterfall, agile, and the critical path method.
What tools and software are most effective for managing pharmaceutical projects?
To manage a complex multi-project environment, you need software that combines project visibility, analytical capabilities, and portfolio balancing. Epicflow is a good example of software like this. Its key feature, EPO, can automatically balance a portfolio of pharmaceutical projects with business value maximization in mind.